REUTERS
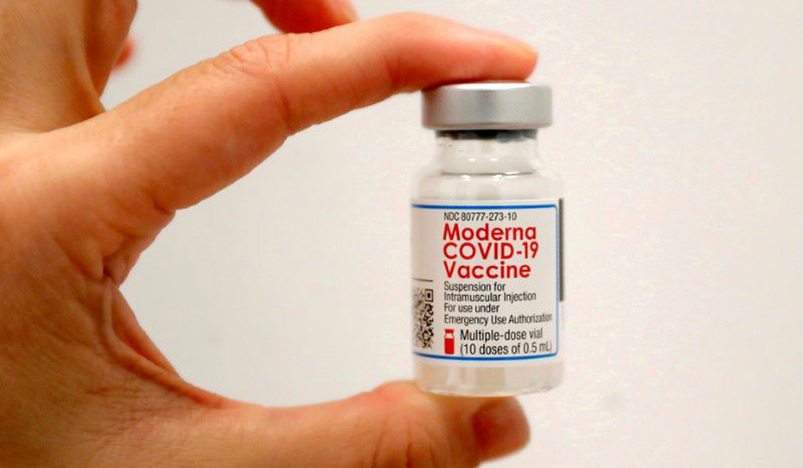
Moderna Inc (MRNA.O) is planning to double the size of an ongoing trial testing its COVID-19 vaccines in children aged six months to less than 12 years.
According to updated details on clinical trial record site clinicaltrials.gov, the study will now enroll an estimated 13,275 participants, nearly double the earlier target of 6,975 participants.
In July, Moderna said it was in discussions with the U.S. Food & Drug Administration to expand the study with an objective to enroll a larger safety database, which increases the likelihood of detecting rarer events.
The company's vaccine, which received its emergency authorization for people aged 18 and older in the United States in December, is currently under an FDA review for use in adolescents.
Rival shot from Pfizer Inc (PFE.N) and its German partner BioNTech (22UAy.DE) was authorized for ages 12 to 15 years earlier this year.
Reporting by Mrinalika Roy in Bengaluru; Editing by Maju Samuel

 By Qatar Day - August 12, 2021
By Qatar Day - August 12, 2021


.jpg)
.jpg)
.jpg)

.jpg)


.jpg)
.jpg)




.jpg)


.jpg)
.png)
.jpg)











Leave a comment