REUTERS
Risk of Bell's Palsy, a type of facial paralysis, is higher after the first dose of Sinovac Biotech Ltd's (SVA.O) COVID-19 vaccine CoronaVac, according to a study published in The Lancet Infectious Diseases journal.
"The beneficial and protective effects of the inactivated COVID-19 vaccine far outweigh the risk of this generally self-limiting adverse event," the study mentioned.
The study conducted in more than 451,000 individuals showed 28 clinically confirmed cases of Bell's Palsy following CoronaVac, compared to 16 cases following Pfizer/BioNtech's vaccine.
"Our findings suggest an overall increased risk of Bell's palsy after CoronaVac vaccination," according to the study.
The study performed in Hong Kong assessed the risk of the adverse event within 42 days following vaccination.
Sinovac did not immediately respond to a request for a comment.
Reporting by Aishwarya Nair in Bengaluru; Editing by Rashmi Aich

 By Qatar Day - August 17, 2021
By Qatar Day - August 17, 2021
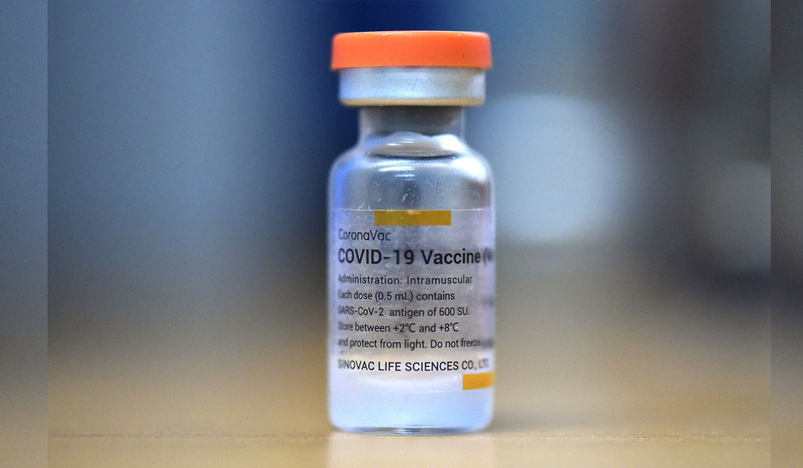

_02-39-2026_11-39.jpg)
_01-34-2026_04-34.jpg)

_01-04-2026_05-04.jpg)

_03-27-2026_08-27.jpg)
.jpg)
.jpg)
.jpg)


.jpg)
.jpg)




.jpg)


.jpg)
.png)
.jpg)











Leave a comment